The role of artificial intelligence (AI) in drug design has fundamentally shifted from a speculative tool to a central pillar of pharmaceutical research and development (R&D). Sino Biological plays a critical role in this evolving landscape by acting as an essential bridge between computational prediction and experimental truth.
Current AI trends in drug development
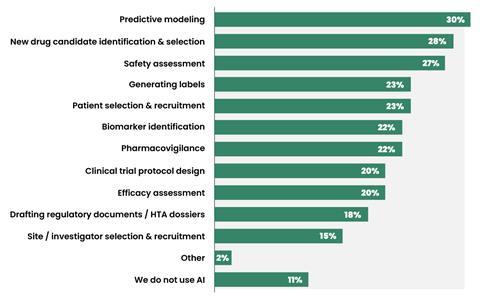
According to an industry survey, approximately 81% of pharma organizations now utilize AI in at least one development program1. Predictive modeling and new drug candidate identification and selection are among the major workflows where AI is being applied in pharma R&D (Figure 1). This trend is backed by staggering financial commitments. Large pharma players, such as Pfizer, Takeda, and AstraZeneca, have ramped up their AI investment in recent years, with a particular focus on drug discovery.
The global AI-powered drug discovery market was valued at approximately $3.1 billion in 2025 and is expected to rise to $4 billion in 20262. Top AI-first biotechs include Isomorphic Labs (Alphabet), Insitro, Insilico Medicine, and Recursion2. These key players are leveraging proprietary platforms and high-value collaborations with traditional pharmaceutical companies to reduce development timelines, lower overall costs, and increase the probability of drug candidate success. For instance, Isomorphic Labs signed AI drug discovery deals worth nearly $3 billion with pharmaceutical giants Eli Lilly and Novartis, and another AI startup Chai Discovery announced a partnership with Eli Lilly to accelerate drug discovery using generative design models3,4.
The role of high-throughput expression in AI design
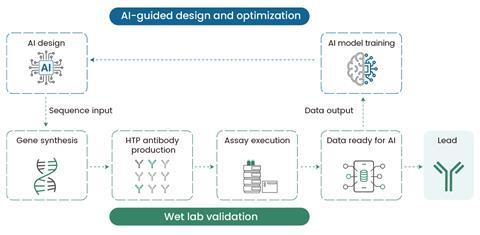
As AI models become increasingly adept at de novo design, the demand for rapid wet lab validation has skyrocketed5,6. Sino Biological’s high-throughput (HTP) antibody production platforms based on dual expression systems–mammalian and cell-free systems–have emerged as transformative tools capable of rapidly producing thousands of AI-designed antibody variants. This enables researchers to move seamlessly from AI-generated sequences to functional antibodies in just days, supporting the fast design-build-test-learn cycles required to refine AI models (Figure 2).
Proven mammalian excellence & rapid cell-free innovation
Traditional mammalian expression systems, such as HEK293 and CHO cell lines, remain the industry gold standard for HTP production of recombinant antibodies, including full-length IgG, VHH, and scFv. Sino Biological leverages deep expertise in HTP gene synthesis, vector construction, and optimized transient antibody expression technology to produce small-scale, high-quality recombinant antibodies, delivering industry-leading throughput (10,000+ antibodies/month) and speed (10 days from gene to antibody). Additionally, immediate access to a catalog of 10,000+ premium recombinant proteins allows for rapid, accurate binding validation of these antibodies, ensuring specificity and high-quality data.
For projects requiring even faster turnaround or involving difficult-to-express proteins in mammalian cells, cell-free protein synthesis (CFPS) systems offer a rapid and efficient alternative7. CFPS, also termed in vitro translation, facilitates the HTP production of target proteins from DNA templates by harnessing the translational machinery outside of living cells (Figure 3). This approach eliminates the speed bottleneck of cell-based systems, slashing production timelines from weeks to hours. At Sino Biological, antibody expression in CFPS systems is completed in just 3 hours, matching the pace of AI-driven discovery. From AI-generated sequences to synthesis, expression, purification, and validation, the entire workflow is seamless, enabling the transition from in silico design to functional antibodies in mere days. Additionally, by bypassing cellular viability constraints, CFPS enables the synthesis of toxic or challenging proteins and the incorporation of non-natural amino acids, fully unlocking the potential of AI-driven design.
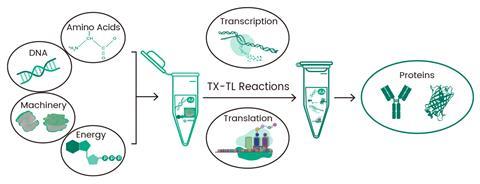 Figure 3. Cell-free protein synthesis process.
Figure 3. Cell-free protein synthesis process.
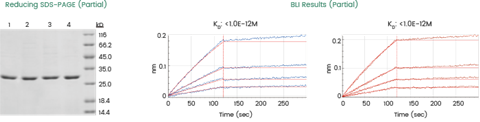
Case study of HTP scFv-His and VHH-His synthesis via CFPS: The HTP capability of Sino Biological’s CFPS platform was recently demonstrated in a project involving over 2,000 AI-designed scFv and VHH sequences (Figure 4). The entire library was synthesized and expressed in parallel, followed by binding affinity analysis via BLI. This integrated HTP workflow–bridging production and characterization–facilitated the efficient identification of promising leads with picomolar affinity and the generation of essential data for further pipeline optimization.
Format: 2,000+ scFv-His and VHH-His antibodies
Expression system: CFPS
Target: Finished expression and affinity analysis with different target proteins by BLI
De-risking antibody discovery through early developability assessment
Early-stage developability profiling is pivotal for de-risking the drug discovery pipeline and streamlining candidate selection8,9. Comprehensive antibody developability evaluation enables the proactive identification and management of potential roadblocks, such as filtering out candidates with suitable binding characteristics but undesirable biophysical properties.
To accelerate candidate triaging and early-stage optimization, Sino Biological provides an integrated platform for HTP antibody developability assessment, encompassing approximately 20 ready-to-use assays to evaluate key attributes such as homogeneity, stability, solubility, and specificity. Flexible assay selection and customization are available to meet specific project requirements. Utilizing this platform, comprehensive developability properties–including thermal stability (nanoDSF/DSC), hydrophobicity (HIC-HPLC/PAIA-HIC), self-association (AC-SINS), polyreactivity (BVP/DNA/Insulin ELISA), and colloidal stability (SMAC-HPLC)–can be thoroughly assessed (Table 1). High-quality, structured data is delivered efficiently, supporting AI/ML model training and accelerating drug discovery campaigns.
Table 1. Antibody developability assays at Sino Biological.
| Category | Assays |
|---|---|
|
Purity |
SEC-HPLC/SDS-PAGE |
|
Titer |
ELISA |
|
Solubility |
DLS |
|
Intact Mass |
LC-MS |
|
Colloidal Stability |
SMAC-HPLC |
|
Thermo Stability |
nanoDSF/DSC |
|
Aggregation |
SEC-HPLC/SEC-MALS |
|
Self-Association |
AC-SINS |
|
Hydrophobicity |
HIC-HPLC/PAIA-HIC |
|
Size Distribution and Aggregation |
DLS |
|
Target Binding |
ELISA/SPR/BLI |
|
Affnity Analysis |
FACS/SPR/BLI |
|
FcγR/FcRn/C1q Binding |
SPR/BLI |
|
Polyreactivity |
BVP/DNA/Insulin ELISA |
|
Cell-Based Assay |
Assay Specific |
Synchronizing discovery: An Integrated HTP platform for AI-driven innovation
As AI-driven innovation accelerates, aligning experimental throughput with the scale of modern discovery is paramount. This is especially critical as antibody pipelines expand, fueling the demand for seamless HTP expression and parallelized assay workflows.
Sino Biological’s integrated antibody production and developability assessment platform streamlines this process through a comprehensive HTP approach, enabling the evaluation of thousands of antibodies within compressed timelines. From AI-designed sequence inputs to robust, high-precision data outputs–this all-in-one HTP platform provides a complete picture of antibody developability profiles, effectively accelerating the transition from initial discovery to advanced therapeutics.
References
1. Norstella. Assessing the impact of AI transformation on pharma R&D. [Online]. Available at: https://www.norstella.com/assessing-ai-transformation-pharma-rd/ (Accessed: 24 March 2026).
2. Global Market Insights. AI in drug discovery market size, share & forecast report 2024–2032. [Online]. Available at: https://www.gminsights.com/industry-analysis/ai-in-drug-discovery-market (Accessed: 24 March 2026).
3. Isomorphic Labs. Isomorphic Labs kicks off 2024 with two pharmaceutical collaborations. [Online]. Available at: https://www.isomorphiclabs.com/articles/isomorphic-labs-kicks-off-2024-with-two-pharmaceutical-collaborations (Accessed: 24 March 2026).
4. TechCrunch. From OpenAI’s offices to a deal with Eli Lilly: How Chai Discovery became one of the flashiest names in AI drug development. [Online]. Available at: https://techcrunch.com/2026/01/16/from-openais-offices-to-a-deal-with-eli-lilly-how-chai-discovery-became-one-of-the-flashiest-names-in-ai-drug-development/ (Accessed: 24 March 2026).
5. Zhu H, et al. Integration of AI and high-throughput technologies in drug discovery. Biology. 2025; 14(9): 1268. https://doi.org/10.3390/biology14091268
6. Li X, et al. Advanced drug delivery systems and AI-driven formulation optimization. Pharmaceutics. 2023; 15(7): 1916. https://doi.org/10.3390/pharmaceutics15071916
7. Wang Y, et al. Monoclonal antibody developability assessment: Challenges and opportunities. Antibodies. 2015; 4(1): 12. https://doi.org/10.3390/antib4010012
8. Chen L, et al. Emerging trends in AI-powered biopharmaceutical R&D. Military Medical Research. 2025; 12: 00764-4. https://doi.org/10.1186/s40364-025-00764-4
9. Nature Biomedical Engineering. Navigating the interface of AI and wet lab automation. Nat Biomed Eng. 2025; 9: 00349-8. https://doi.org/10.1038/s44222-025-00349-8



















No comments yet